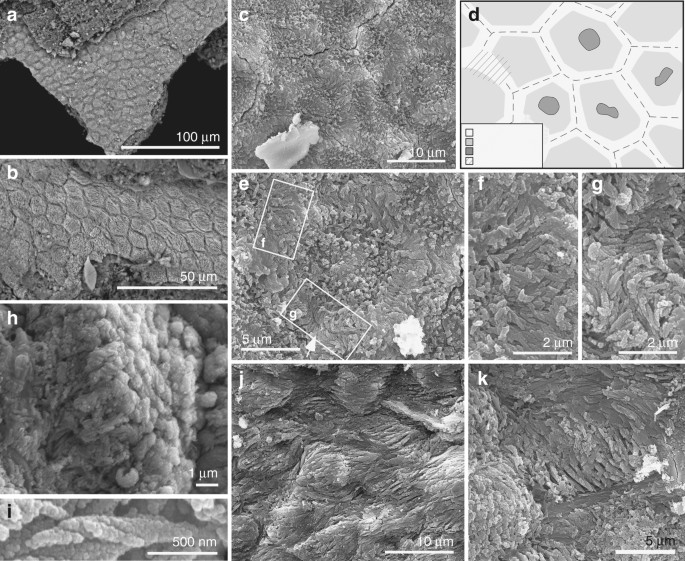
.
Assim se chama o mais antigo mamífero conhecido para as terras
brasileiras. Viveu entre 87 milhões e 70 milhões de anos atrás no fim da
era Mesozoica, onde hoje é o noroeste do Estado de São Paulo. Trata-se
do único mamífero brasileiro que, sabe-se até o momento, conviveu com os
dinossauros.
,
professor da Faculdade de Filosofia, Ciências e Letras de Ribeirão
Preto (FFCLRP) da Universidade de São Paulo, que teve apoio de colegas
da Universidade Federal de Goiás, da Universidade Estadual de Campinas
do Museu de La Plata e do Massachusetts Institute of Technology.
Brasilestes foi descrito a partir de um único fóssil: um dente pré-molar de 3,5 milímetros. “O dente de
Brasilestes é pequeno e se encontra incompleto, pois lhe faltam as raízes”, disse a paleontóloga
Mariela Cordeiro de Castro, a primeira autora do trabalho que acaba de ser publicado na
Royal Society Open Science.
“Pequeno mas nem tanto. Apesar de só ter 3,5 milímetros, o dente do
Brasilestes
é três vezes maior do que quase todos os dentes conhecidos de mamíferos
do Mesozoico. No tempo dos dinossauros, a maioria dos mamíferos tinha o
tamanho de camundongos.
Brasilestes era bem maior: do tamanho de um gambá”, disse Castro.
O nome da nova espécie homenageia o roqueiro inglês David Bowie, morto em janeiro de 2016, um mês após a descoberta do fóssil.
Brasilestes stardusti faz alusão a Ziggy Stardust, ou Ziggy “poeira estelar”, personagem vindo do espaço que Bowie criou para uma canção de 1973.
O trabalho contou com apoio da FAPESP e integra o Projeto Temático
“A origem e irradiação dos dinossauros no Gondwana (Neotriássico - Eojurássico)”, coordenado por Langer.
O dente fossilizado encontrado em rochas da Formação Adamantina que
afloram no meio de um pasto na fazenda Buriti, em General Salgado (SP).
“Estávamos visitando afloramentos mesozoicos quando
Júlio Marsola [outro
membro da equipe] teve um olhar de lince e avistou um dente minúsculo
aflorando na superfície da rocha”, disse Castro, professora na
Universidade Federal de Goiás, em Catalão. “Brinco que, naquele dia, ele
gastou a sua quota de descobertas extraordinárias para toda uma vida – e
olha que ele nem estuda mamíferos, mas dinossauros.”
“Quando penso que alguns grupos de pesquisa chegam a peneirar uma
tonelada de sedimento pra achar um único fragmento de mamífero do
Mesozoico, acredito que Mariela tenha razão, e que eu tenha de fato
gastado toda minha sorte. Mas espero sinceramente que não”, brincou
Júlio Marsola, pesquisador da FFCLRP-USP.
“Os depósitos de General Salgado são bem conhecidos. De lá já saíram
vários crocodilos mesozoicos. O afloramento em particular onde achei
Brasilestes
é interessante, com dezenas de fragmentos de cascas de ovos daqueles
crocodilos. Ao me debruçar em uma parte do afloramento para ver o que
possivelmente tinha de cascas de ovos, me deparei com o dentinho. Se
permanecesse mais alguns dias exposto, a chuva levaria embora”, disse
Marsola.
“Quando percebi algo como a base das duas raízes do dente [as raízes
em si estão quebradas], achei que fosse um mamífero. Depois de analisar
em laboratório, tivemos a certeza de que se tratava de um mamífero”,
disse.
Placentário no deserto Botucatu
À primeira vista, um mero dente de 3,5 milímetros – ainda por cima
incompleto – pode parecer muito pouco para descrever uma nova espécie de
mamífero. Não é. É comum se ver mamíferos extintos descritos com base
no estudo de um único dente.
Isso se deve ao fato de que o material que constitui os dentes é o
mais resistente do esqueleto dos mamíferos, pois precisa resistir ao
desgaste provocado pela mastigação durante toda a vida do animal –
diferentemente dos peixes e de inúmeros répteis, por exemplo, que trocam
dentes ao longo da vida.
Por serem muito duros e resistentes, os dentes são os restos do
esqueleto mamífero com maiores chances de preservação após a morte do
animal. Com frequência, são os únicos a permanecer intactos por tempo
suficiente para ter chance de fossilizar. Há muitos exemplos de espécies
extintas de mamíferos, inclusive de ancestrais do ser humano, descritas
com base em um único canino ou molar.
A singularidade e a incompletude do pré-molar de
Brasilestes
impedem que os pesquisadores distinguam com absoluta confiança a qual
grupo de mamíferos a espécie pertencia. Sabe-se que o dente saiu da boca
de um tério, ou seja, de um membro do grande grupo que congrega
marsupiais e placentários.
Muito embora não haja evidências suficientes para sustentar a inclusão de
Brasilestes em um grupo ou outro, ainda assim os especialistas têm a impressão, sem afirmar categoricamente, de que
Brasilestes teria sido placentário. E isto é completamente inusitado.
Há atualmente três grandes grupos de mamíferos: placentários,
marsupiais e monotremados. Os três grupos evoluíram no Mesozoico. Porém,
naquele tempo, estavam longe de ser as únicas formas de mamíferos.
Havia diversos outros grupos, como os multituberculados comuns no
hemisfério Norte, e grupos típicos do hemisfério Sul, como
meridiolestídeos e gonduanatérios – alusão ao Gondwana, o antigo
supercontinente austral que reunia América do Sul, África, Índia,
Austrália e Antártica.
Desde o início dos anos 1980, quando começaram a ser achados os
primeiros fósseis de mamíferos mesozoicos na Patagônia argentina ( hoje
são conhecidas cerca de 30 espécies, as únicas do continente até o
anúncio de
Brasilestes), jamais se descobriu nada remotamente parecido com o dentinho brasileiro.
“Quando mostrei o fóssil de
Brasilestes ao paleontólogo
Edgardo Ortiz-Jaureguizar, do Museu de La Plata, ele ficou muito
surpreso, afirmou nunca ter visto nada parecido, e correu para mostrar o
fóssil a outro especialista daquela instituição, Francisco Goin”, disse
Castro. “A reação foi idêntica. Goin afirmou que
Brasilestes não se parecia com nenhum outro mamífero mesozoico achado na Argentina – vale dizer, na América do Sul.”
Entre as cerca de 30 espécies de mamíferos mesozoicos argentinos, há
meridiolestídeos, gonduanatérios e até mesmo, suspeita-se, um ou outro
multituberculado. Mas nenhum marsupial nem placentário. Os fósseis
desses dois grupos só começam a surgir no registro fóssil sul-americano
após a extinção em massa que exterminou com os dinossauros há 66 milhões
de anos, evento que põe fim ao Mesozoico e dá início à era atual, o
Cenozoico.
Até a descoberta de Brasilestes, os únicos vestígios de
mamíferos mesozoicos no Brasil se resumiam a centenas de trilhas e
pegadas deixadas por criaturas desconhecidas que caminhavam há 130
milhões de anos sobre as dunas do antigo deserto Botucatu, que cobria o
interior de São Paulo. A superfície de tais dunas, solidificada, chegou
aos nossos dias sob a forma de lajes de arenito onde se veem as antigas
pegadas.
Em 1993,
Reinaldo José Bertini,
professor na Universidade Estadual Paulista (Unesp), em Rio Claro,
chegou a anunciar a descoberta de um fragmento de mandíbula de mamífero
portando um único dente – e muito menor que o pré-molar de
Brasilestes. Bertini, porém, não publicou um estudo minucioso daquele fóssil, nomeando assim uma nova espécie.
“Temos assim que
Brasilestes, além de ser o primeiro mamífero
descrito para o Mesozoico brasileiro, também é um dos poucos mamíferos
mesozoicos descobertos em regiões mais centrais da América do Sul, dado
que os fósseis argentinos foram achados em formações geológicas na
Patagônia, portanto no sul do continente”, disse Langer.
“Não bastasse isso,
Brasilestes difere de tudo o que se
encontrou antes dele, indicando que possivelmente mamíferos placentários
habitavam a América do Sul entre 87,8 milhões e 70 milhões de anos
atrás”, disse Langer.
O mais inusitado é que o mamífero mesozoico com pré-molares mais parecidos com os do
Brasilestes viveu do outro lado do mundo, na Índia, entre 70 milhões e 66 milhões de anos atrás.
Chamava-se
Deccanolestes. Nenhuma outra criatura do registro fóssil mundial guarda tamanha semelhança com
Brasilestes.
O que dois membros da mesma linhagem estariam vivendo em regiões tão
afastadas e não conectadas? Há cerca de 100 milhões de anos, ao mesmo
tempo em que a América do Sul e a África acabavam de se separar
definitivamente com a abertura do Atlântico Sul, a Índia cortava suas
amarras do Gondwana e começava a vagar pelo oceano Índico.
Isso implica que, até pelo menos 100 milhões de anos atrás, ancestrais de
Brasilestes e
Deccanolestes povoavam o supercontinente Gondwana. Ou seja, a linhagem de
Brasilestes e
Deccanolestes
é muito mais antiga do que a idade de seus respectivos fósseis: entre
87 milhões e 70 milhões de anos para Brasilestes, e entre 70 milhões e
66 milhões de anos para
Deccanolestes.
“A descoberta de
Brasilestes suscita muito mais dúvidas do que respostas sobre a biogeografia dos mamíferos mesozoicos sul-americanos. Graças a
Brasilestes, descobrimos que a história dos mamíferos de Gondwana é mais complexa do que se supunha”, disse Langer.
Disso podem brotar novas hipóteses e surgir novas linhas de
investigação. Quem sabe, por exemplo, futuras pesquisas disparadas a
partir da descoberta de
Brasilestes não venham a desvendar a
origem ainda desconhecida de um grupo típico da América do Sul: os
xenartros, a ordem dos tatus, tamanduás e preguiças? Por sinal, a
especialidade de Mariela Castro é a história evolutiva dos xenartros.
“Um traço interessante do pré-molar de
Brasilestes é a espessura do seu esmalte, superfino, com apenas 20 mícrons. O esmalte de
Brasilestes
é o mais fino para os dentes de quaisquer mamíferos do Cretáceo no
registro fóssil.
A maioria dos dentes de mamíferos mesozoicos tem
esmalte com espessuras entre 100 e 300 micrômetros”, disse Castro.
“Entre os xenartros, conhecem-se dezenas de espécies viventes e
centenas de extintas. Somente três possuem esmalte. Dois tatus extintos e
o tatu-galinha (
Dasypus novemcinctus), o único xenartro vivente com esmalte.
A microestrutura do esmalte do pré-molar de
Brasilestes e dos pré-molares do tatu-galinha é muito parecida”, disse.
Segundo a paleontóloga, “a evidência do relógio molecular sugere que a
linhagem dos xenartros surgiu há pelo menos 85 milhões de anos. Porém
os fósseis mais antigos de tatus, achados no Rio de Janeiro, têm cerca
de 50 milhões de anos”.
Embora seja bastante sugestivo imaginar
Brasilestes como um antigo xenartro, ainda é muito cedo para fazer qualquer afirmação nesse sentido.
“Embora a idade e a proveniência de
Brasilestes coincidam com
hipóteses moleculares para a origem dos xenartros, a inferência da
afinidade taxonômica é prematura diante das diferenças morfológicas
entre o dente de
Brasilestes e os dentes de tatus”, disse.
Langer concorda. “Só temos um único fóssil de
Brasilestes. Seu registro é por demais incompleto para extrair novas conclusões”, disse.
Como nunca antes de
Brasilestes foram encontrados fósseis de
mamíferos mesozoicos no Brasil, isso pode significar que tais fósseis
são raros ou de difícil preservação. “Quem sabe um dia encontremos novos
fósseis de
Brasilestes que ajudem a entender melhor sua história. Pode levar décadas”, disse Langer.
O artigo
A late Cretaceous mammal from Brazil and the first radioisotopic age for the Bauru Group,
de Castro MC, Goin FJ, Ortiz-Jaureguizar E, Vieytes EC, Tsukui K,
Ramezani J, Batezelli A, Marsola JCA e Langer MC, está publicado pela
Royal Society Open Science em
http://rsos.royalsocietypublishing.org/lookup/doi/10.1098/rsos.180482.